ITEST™CARBOHYDRATES analyses for pome fruit
by Dr Elmi Lötze (Head of ITEST™CARBOHYDRATES and ITEST™LEAF) and Wilmé Brown (Plant Physiologist)
Biological indicator for management decisions
The ITEST™CARBOHYDRATES service is now also available for pears and apples. Although the impact of carbohydrates on pome fruit cultivation has been investigated locally for many years, a commercial service making it possible to utilise this physiological indicator for the purpose of making management decisions, has never been available.
With the ITEST™CARBOHYDRATES service, carbohydrate leaf, shoot and root samples are taken during critical action times (CATs) to measure the tree’s energy status and manage the vegetative and reproductive development accordingly. This communication will focus specifically on root carbohydrates.
Root carbohydrates
Accumulation
Although all permanent organs in apple and pear trees fulfil a storage function for nutrients and carbohydrate reserves, the majority of the starch usually accumulates in the roots.
Starch accumulation in the roots follows a dynamic seasonal pattern that coheres with the phenological stages of the tree, thereby ensuring that the energy status of the tree remains in balance. Starch accumulates mainly at the end of the growing season, after shoot growth cessation and harvest (Stassen, 1984), when the plant organs’ sink strength for carbohydrates above ground decreases and leaves can focus primarily on exporting sugar to build reserves in the permanent tissues (Figure 1).
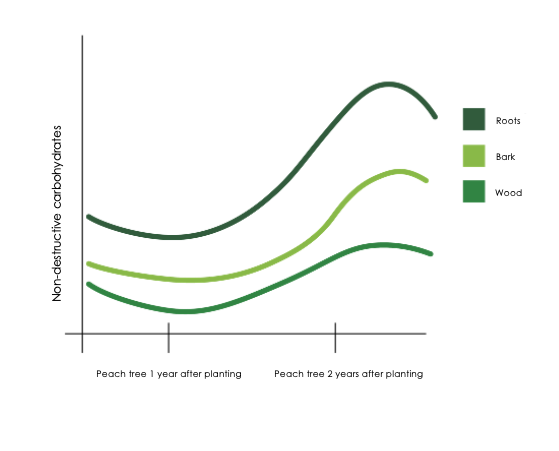
Figure 1: Seasonal changes in the carbohydrate fractions in a young peach .
Leaf drop and dormancy
After leaf drop, starch levels gradually decrease during winter due to respiration of especially shoots and buds, to maintain the metabolism of the dormant tree in the absence of the photosynthesizing organs – the leaves (Figure 2). The reserve status of permanent structures (such as roots) during this leafless phase will therefore contribute significantly to the energy status of the tree and processes such as cell division of buds, during dormancy.
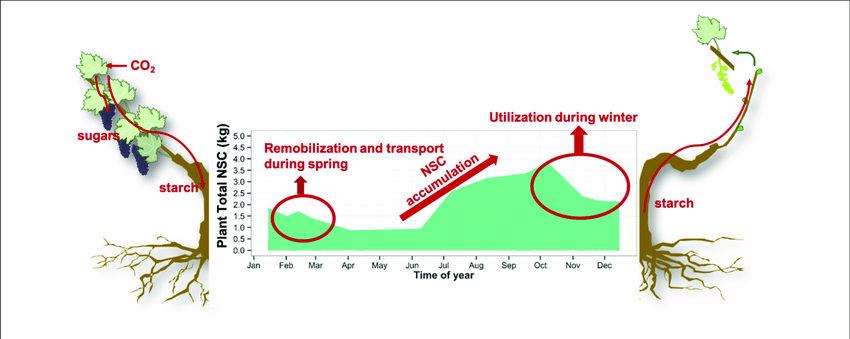
Figure 2: The seasonal course of carbohydrate accumulation and translocation in deciduous crops from the beginning of spring and leaf development, to winter and dormant, leafless period.
Dormant buds continue to develop during winter and cell division and differentiation (the formation of the flower parts into reproductive flower buds) also require energy – which must now come from reserves (Figures 3 and 4).
In addition, carbohydrates are required for the formation of active, white root tips during the peak white root flush – May to August (bearing apple trees in Western Cape) (Lötze et al., 2020), when no leaves are present to provide sugar.
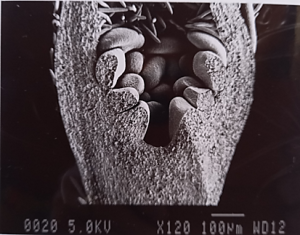
Figure 3: Scanning electron microscope photo of the development of a Royal Gala flower bud in July.
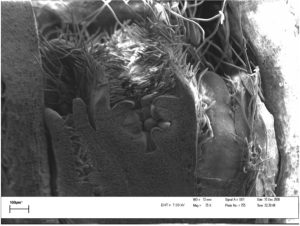
Figure 4: Apple reproductive bud in June.
Adequate carbohydrate levels in roots are critical
Root growth initiation is responsible for the production of essential hormones such as cytokinin, which is required above ground for bud break and shoot growth and, together with an increase in abscisic acid, stimulates reproductive development. Therefore, suboptimal root starch levels (reserves) will reduce vegetative development, gibberellic acid levels and the subsequent development of bearing positions and thus, yield will decline as well (Bower et al., 1990).
During spring, a rapid decrease in the starch levels of storage organs occur from bud break and subsequent vegetative growth stages, to meet the high energy demand for processes that cannot yet be provided by photosynthesis.
Starch in the roots, stem and shoots is then converted into sugar and transported to the developing buds. Local sugar analyses of avocado flowers by Agri Technovation have indicated that flowers have particularly high sugar levels. An avocado flower contains approximately 23% sugar, 4% starch and 73% water and minerals, confirming results from carbohydrate analyses of apple flowers by Vemmos (1995).
Sufficient reserves
The timeous accumulation of carbohydrates and availability of sugar at bud break are therefore essential for even bud break during spring (Stassen, 1980; 1984). In deciduous crops, the only opportunity for this is the period from shoot growth cessation to the start of leaf drop, when the canopy is fully developed and photosynthesis optimal, and starch is able to accumulate in the permanent tissues. Adverse climatic conditions and pest or disease outbreaks during late summer could have a significant adverse impact on carbohydrate accumulation (Loescher et al., 1990).
To determine how a post-harvest fertilisation strategy should be adapted to ensure an optimal carbohydrate status during winter, a carbohydrate sample of leaves and roots can be taken. Fertiliser recommendations can thus be adjusted in time, along with irrigation, for carbohydrate accumulation. A low carbohydrate status during this stage can also be partially addressed by fruit thinning during winter pruning.
The effect of winter and the potential crop load on the available sugar at bud break can be determined with a carbohydrate sample at the end of winter. Thus, corrective actions to be made from flowering onwards can be planned in a timely manner.
Critical action times at pome fruit and management actions to address carbohydrate index
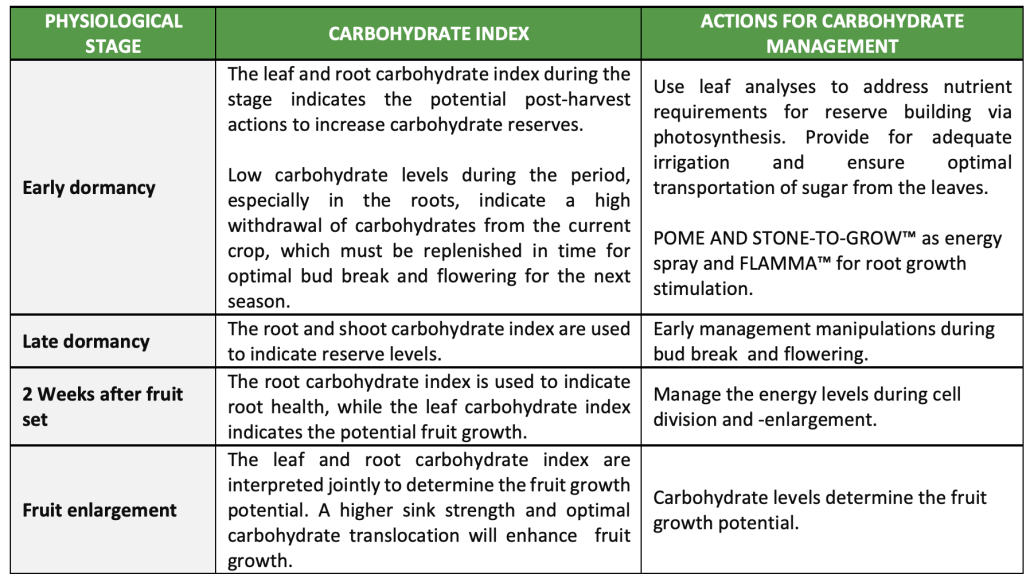
Summary
Root health is of great importance for optimal carbohydrate storage capacity, the critical uptake of some nutrient elements and the synthesis of hormones that drive further growth and development. Determining and responding to the carbohydrate levels of roots early can therefore result in good fruit quality and optimal yield.
References:
- Bower et al., 1990. Interaction of plant growth regulators and carbohydrate in flowering and fruit set. Acta Hort. 275:425-434.
- Loescher et al., 1990. Carbohydrate Reserves, Translocation, and Storage in Woody Plant Roots. 25(3): 274-281.
- Stassen, 1980. A study on the carbohydrate and nitrogen metabolism in Prunus Persica. PhD dissertation, Stellenbosch University.
- Stassen, 1984. Seisoensveranderinge in die koolhidraatinhoud van jong appelbome. Afr. J. Plant Soil 1984, 1(3): 92-95.
- Vemmos, 1995. Carbohydrate changes in flowers, leaves, shoots and spurs of Cox’s Orange Pippin’ apple during flowering and fruit setting periods. Hort. Sci.70(6):889-900.
- Lötze et al., 2020. White root tip dynamics of bearing apple trees in a Mediterranean climate, South Africa. Acta Hort. 1281, 347-354

